Basic Information
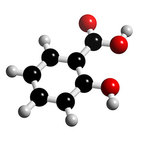
- A beta hydroxy acid
- Comes from the bark of willow trees
- Formula of C7H6O3
- Molecular mass of 138.1 g/u
- Melting point of 159 C
- Boiling point of 211 C
- Density of 1.4 g/cm^3
- Slightly soluble in water
- pH of 2.4
- Four double bonds; 3 Carbon to Carbon and 1 Carbon to Oxygen
- Percent compisition: Carbon 60.87%, Hydrogen 4.38%, Oxygen 34.75%
- Used in wart removal, the treatment of dermatitis, psoriasis and acne.
History

Salicylic acid was first noticed by ancient greeks as a fever reducer by chewing on the bark of willow trees. It wasn't used as a skin treatment until the 1900s. Acetylsalicylic acid was found when Charles Gerhardt replaced the hydroxide in salicylic acid with a new compound in 1853. This would later help create aspirin.
